Japanese scientists have made important progress in solving the problem of human infertility. They used a female blood cell, human-induced stem cells (hPGCLCs), to create human oogonia cells in a laboratory dish. This research is of great significance, meaning that human babies may be born in the laboratory in the future, thus changing the pattern of human reproduction.
However, scientists have only created primitive germ cells, are not mature eggs, and cannot be used for fertilization and embryos. Previously, researchers have used the tail cells of mice to make eggs and insemination, resulting in fertile pups. Therefore, outside scientists believe that the research is expected to achieve human “in vitro gametes” in the future – a method of making eggs and sperm in a culture dish.
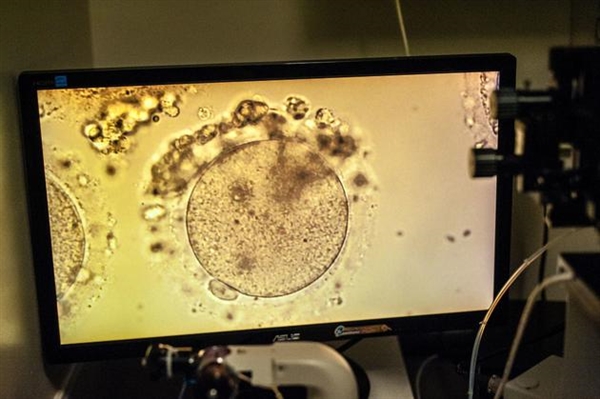
“It’s only a matter of time before we achieve the same in humans. We haven’t been completely successful, but we can’t deny that this is a great step,” said Eli Adashi, former head of the School of Medicine and Biological Sciences at Brown University who was not involved in the study. “Considering how difficult it is for humans, this new study has broken this situation to some extent.” In February 2016, Chinese scientists succeeded in obtaining functional sperm in vitro for mice. Male infertility brings hope.
For many years, scientists have been trying to apply the regenerative potential of stem cells to different purposes, hoping they can be used to regenerate damaged brain cells in patients with cardiomyocytes or Parkinson’s disease. Studies more than a decade ago found that ordinary skin cells or blood cells can also be recoded into stem cells and have the ability to develop into any type of tissue in the body, which is one of the most attractive research directions in the biomedical field.
The new study, led by stem cell research expert at Kyoto University in Japan, led to the creation of stem cells from human blood cells and then directs them to develop into primordial germ cells. His team placed the cells in a petri dish containing mouse ovary cells, which remained active for 4 months and developed into oogonia cells. In humans, oogonia is the precursor of mature egg cells that appear in the first trimester of pregnancy.
“I think this is an important step, but it is one of the few steps that must be experienced before using stem cells to make usable eggs and sperm,” said Stanford Center for Legal and Biological Sciences, End of Sex and Human Reproduction. “This is farther than any previous research on human egg cells, but it is not an egg,” said Henry Greeley, author of the book.
Many scientists believe that creating mature human eggs in the laboratory is only a matter of time, and what is even more difficult is the series of basic safety and ethical issues that follow. Toshi Shioda, a stem cell biologist at Harvard Medical School, points out that even babies born with this technology face concerns about possible cancer or other diseases, even if they overcome technical challenges.
After security issues are resolved, social and ethical issues will begin to multiply. If germ cells can be made from cheek cells, will anyone become a parent without knowing it? If the demand for eggs ends, will the female’s biological clock be reversed or disappeared? The difficulty in making an egg is reduced, will it make in vitro fertilization a routine operation? Also, if it is easier to produce eggs, will parents be more frequently screened for genetic diseases?
Many scientists say that it is time to discuss these issues, and it is imperative to educate the public about how to supervise. Eli Adashi said: “When we see the next scientific paper…maybe it is not the best time to discuss. There will be potential opposition and hostilities, driven by political, religious and other factors. “This will put the technology in a state of stagnation.” He added that this technology may be the biggest breakthrough in human reproduction since the birth of IVF technology.
Toshi Shioda pointed out that the direct application of this technology will come earlier than any distant “reproductive revolution.” If researchers have a large number of developing germ cells, they can systematically test and understand how drugs or environmental exposure affect human eggs. Scientists can better understand the effects of chemotherapy, toxic chemicals, or nuclear power plant radiation on germ cells. Saito Tatsuhisa said his next goal is to develop a way to further culture oogonia cells, perhaps by incubating them with human ovarian cells instead of using mouse ovarian cells.
Although the study raised ethical issues, it also caused a lot of controversies when in vitro fertilization technology first appeared. Scientists working in the field say they often receive e-mails from people who are unable to conceive, expressing hope that the technology will be applied as soon as possible.
“If we can do this if the technology works and is safe, then hundreds of thousands or millions of couples in the United States today can have genetically-related babies,” Henry Greeley said. There is no obvious reason to say that this doesn’t work. However, the basic understanding of biology tells me that there will always be things that surprise us.”
Source: news.mydrivers.com
Related Posts
Japanese scientists create human oogonia cells in a Vitro
If one day we discover the aliens, what will humans do?
By 2050, you may achieve Immortality and stay alive forever in a body of Robot